Welcome to the OMA Blog
The Obesity Medicine Association Blog is the leading industry hub for obesity medicine. Find the latest research, expert insights, and practical tips to tackle the multifaceted disease of obesity. Hear from OMA Outreach Committee members, OMA Board members, and more to gain a deeper understanding of the complex factors influencing obesity and explore innovative approaches to prevention, treatment, and long-term management. Join a community of healthcare professionals, researchers, and individuals passionate about combating obesity.
View by Category

Understanding Acquired Hypothalamic Obesity
The hypothalamus may be small, but its influence on human physiology is profound. As a central regulator of neuroendocrine function, it plays a critical role in maintaining homeostasis—coordinating hunger and satiety, energy expenditure, sleep–wake cycles, thermoregulation, thirst, and fatigue.1-4 When this finely tuned system is disrupted, the downstream clinical consequences can be severe and lifelong.

Pediatric Research Update: Physical Activity Patterns and Associations in Infants and Toddlers
The most increase in physical activity (PA) was found to be from infancy to 24 months and then tended to plateau. Greater screen time exposure was associated with a slower rate of PA increase between 6 and 24 months of age. Based on these findings, clinicians should consider integrating PA anticipatory guidance into well-child visits beginning in early infancy, with attention to screen time limits and opportunities for active play. In their original article, Longitudinal Change in Physical Activity in Children 6 to 36 Months of Age , Pate and colleagues not only present the developmental pattern for PA in this age group but also highlight factors that may hinder or promote PA development. Interestingly, screen time was associated with higher PA levels at 6 months but with a slower rate of PA growth by 24 months, suggesting that the negative impact of screen exposure on physical activity accumulates over time rather than appearing immediately. Longitudinal Change in Physical Activity in Children 6 to 36 Months of Age.

CMS Announces Changes to Medicare Coverage of GLP-1 Medications for 2027
Medicare coverage of GLP-1s for weight loss indications will occur under the Medicare GLP-1 Bridge Model, rather than the BALANCE Model, for 2027. Eligible Medicare beneficiaries will continue to have access to covered GLP-1 medications nationwide with a fixed $50 copay through the extended Bridge Model. Following previous announcements that Medicare would cover GLP-1 medications through Part D plan sponsors in 2027 under the Better Approaches to Lifestyle and Nutrition for Comprehensive hEalth – or BALANCE – Model , the Centers for Medicare and Medicaid Services (CMS) recently provided updated guidance indicating that the BALANCE model would not launch for Medicare in 2027. The extension of the Bridge Model will enable eligible Part D beneficiaries across the country to access covered GLP-1 medications through December 2027, regardless of their Part D plan sponsors’ participation decisions.

Pediatric Research Update: Obesity and Mental Health in Childhood Adolescence
Article Summary This scoping review highlights the complex, bidirectional relationship between pediatric obesity and mental health, emphasizing depression, psychosocial mediators, and the importance of integrated behavioral and medical interventions in pediatric obesity care. Read the Full Article Article Review Childhood and adolescent obesity is increasingly recognized as a chronic, relapsing condition with significant mental and behavioral health implications. In Obesity and Mental Health in Childhood and Adolescence: A Scoping Review of Recent Scientific Evidence , Morales-Suárez-Varela et al. review recent literature examining the relationship between pediatric obesity and mental health outcomes, with particular attention on depressive symptoms. This review reinforces the importance of addressing mental health and providing mental health treatment as a core component of pediatric obesity care rather than adjunctive care alone. This scoping review synthesizes evidence published over the last decade and highlights the complex, bidirectional relationship between obesity and mental health.

The Latest Obesity Research Findings and Their Impact on Patient Care
Explore the most impactful obesity research, covering breakthroughs in pharmacotherapy, evolving diagnostic criteria, and new bariatric surgery data.
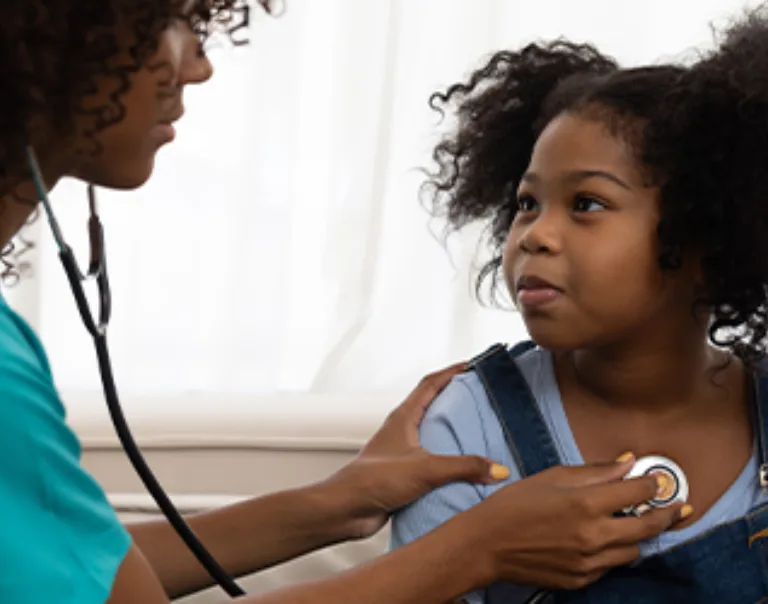
Pediatric Research Update: Pediatric Metabolic and Bariatric Surgery and Antiobesity Medications
Article Summary Treatment Across the Care Continuum in Adolescent Obesity Management: This article reviews the complexity of treating adolescents affected by obesity using antiobesity medications and metabolic bariatric surgery, the potential role of concurrent versus singular treatment strategies and highlights and the need for further research to define optimal timing of these therapies in the adolescent population. Read the Full Article Article Review The treatment landscape for adolescents affected by obesity in the United States has changed dramatically over the past few years. This article highlights the growing impact of adolescent obesity, current information, epidemiology, unique features of adolescent obesity, barriers to care and reviews current treatment options, including antiobesity pharmacotherapy and metabolic bariatric surgery (MBS). Expanding pediatric obesity programs that offer comprehensive care, including lifestyle interventions, anti-obesity medications, and adolescent MBS may help optimize treatment strategies and improve long-term outcomes for adolescents with obesity. Reframing obesity treatment in the adolescent population using a chronic disease model would shift the perspective from viewing treatment as a one-time intervention to recognizing it as part of a continuous, long-term care process. Adopting this model may improve the sustainability of weight management, support the prevention and treatment of weight regain, and promote better long-term health outcomes for adolescents living with obesity. Implications for adult and pediatric obesity specialists include advocating for patients access to evidence-based care, including treatment for adolescents affected by obesity and its associated comorbidities.

Obesity Medicine Association Launches AI-Powered Patient Simulation Tool to Transform Obesity Care Conversations
New “Treating Obesity First” initiative introduces OMAr, an interactive training tool designed to help clinicians practice real-world patient discussions

Top Weight Loss Medications
Discover the most effective weight loss medications approved by the FDA. Comprehensive guide on safety, dosage, and efficacy for 2025.

Tirzepatide vs Semaglutide: A Comprehensive Comparison for Providers
Weight loss medications have undergone a sea change in the past several years. With the rise of incretin therapies, two names have emerged as leaders: tirzepatide and semaglutide. They share many similarities. The frequency with which we hear these two drug names bandied about, along with their brand names — Mounjaro/Zepbound and Ozempic/Wegovy, respectively — can lead to confusion.
