March 12, 2026
How Next-Generation Science Is Improving Health and Redefining Obesity Care
Share this post

Sponsored Content: This blog post was sponsored and submitted by Viking
Obesity is no longer a looming public health threat—it is a full-scale epidemic. More than 40% of U.S. adults live with obesity,1 driving increased risk of type 2 diabetes, cardiovascular-kidney metabolic disease, and reduced quality of life and life expectancy. Long treated as a lifestyle failure, obesity is now clearly recognized as a chronic, biologically regulated disease governed by complex hormonal and neurologic pathways that actively resist weight loss.2,3This growing understanding of obesity biology has shaped how professional societies now define best practice
What the New Guidelines Say: Treat Obesity First
In response, leading professional societies have published updated,4,5 evidence-based guidelines that fundamentally shift obesity care. The message is consistent and clear: treat obesity early, treat it effectively, and treat it as the primary disease. These guidelines emphasize that pharmacotherapy, including GLP-1–based agents, should be considered early alongside lifestyle modification, with the goal of improving overall health outcomes rather than weight alone. Lifestyle interventions remain essential, but are now framed as supportive care rather than prerequisites.4-6 As with cardiology and diabetes, early, guideline-directed treatment of obesity offers an opportunity to change long-term health trajectories—for patients and for public health.
Next-Generation NuSH Therapies: From GLP-1s to Next-Generation NuSH Therapies
The rapid evolution of obesity pharmacotherapy reflects a deeper understanding of the biology that governs appetite, energy balance, and metabolic health.2 While GLP-1 receptor agonists have established an important foundation for obesity treatment,4,5 emerging Nutrition-Sensitive Hormone (NuSH) therapies are being developed to further refine this approach by engaging multiple complementary metabolic pathways.
One such investigational therapy is VK2735, a dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist currently in clinical development.7 In the phase 2 VENTURE study VK-2735 demonstrated substantial weight loss after 13 weeks of oral dosing, with mean reductions of up to 12.2% from baseline. Weight loss was progressive across doses, with statistically significant differences versus placebo observed as early as Week 1 at doses above 15 mg. Up to 97% of participants achieved ≥5% weight loss and 80% achieved ≥10%, compared with 10% and 5% with placebo.7(Figure 1)
Figure 1. Phase 2 VENTURE trial design and key efficacy outcomes with VK2735 in participants with obesity or overweight with at least one weight-related comorbidity.
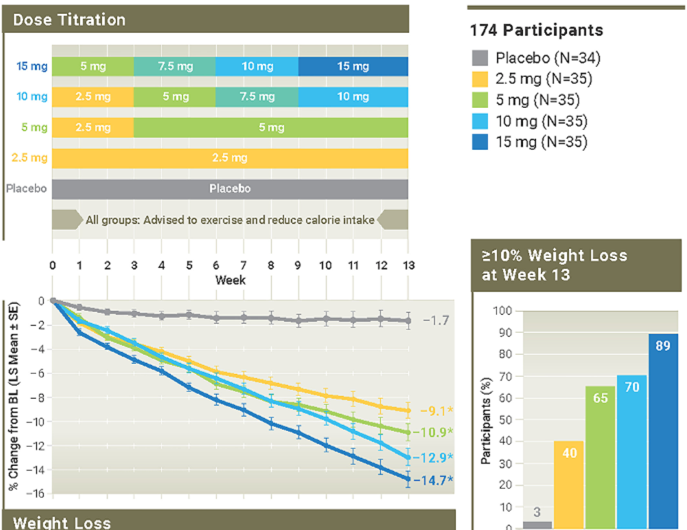
Beyond weight reduction, VK2735 demonstrated favorable metabolic effects. Among participants with prediabetes at baseline, 78% achieved normoglycemia by Week 13, and similar improvements were observed among individuals meeting criteria for metabolic syndrome, highlighting the potential for early cardiometabolic risk modification.7 VK2735 was generally well tolerated, with mostly mild to moderate, transient adverse events—primarily gastrointestinal and consistent with the incretin class. An exploratory maintenance cohort suggested weight loss could be sustained at lower daily doses following initial higher dosing.
Encouraging results from the phase 2 VENTURE study helped shape the ongoing VANQUISH phase 3 program, which is now evaluating VK2735 in larger patient populations. The program includes two randomized trials—VANQUISH-1 in adults with obesity and VANQUISH-2 in adults with obesity and type 2 diabetes—assessing once-weekly VK2735 over 78 weeks, with percent change in body weight as the primary endpoint.8,9 Each study also includes a one-year extension phase.
Ongoing studies are also exploring alternative dosing strategies—including monthly subcutaneous dosing and both daily and weekly oral dosing—to evaluate whether VK2735 could support flexible treatment approaches, including potential transition from injections to oral therapy for long-term weight maintenance.
Looking Forward: Treating Obesity First Through Next-Generation Science
More broadly, innovative next-generation science drugs like VK-2735 reflect a growing shift in obesity medicine toward treating obesity first—addressing the underlying biology of weight regulation rather than focusing solely on downstream complications. As next-generation science continues to expand the therapeutic landscape, clinicians are increasingly equipped to intervene earlier, individualize care, and improve both health outcomes and quality of life for people living with obesity. Together, evolving clinical evidence and updated guidelines underscore the potential of next-generation, biology-based therapies to meaningfully improve life and health across the cardiometabolic disease spectrum.
Sponsor Call-Out
VK2735 is an investigational therapy in clinical development.
- Join us for the Viking sponsored panel discussion Patient Centered Obesity Risk Assessment Approaches on Saturday, March 28, from 6:30 to 8:00 p.m hosted by Harold Bays, MD and John Ostrominski, MD at the Marriott New Orleans Warehouse Arts District, 859 Convention Center Boulevard: (Register Here).
- Viking will be hosting additional educational events focused on integrating obesity treatment into cardiovascular care pathways, and we would be delighted for you to join a future session (Click Here).
-
- Centers for Disease Control and Prevention. Adult obesity facts. Updated May 2024. Accessed February 12, 2026. https://www.cdc.gov/obesity/data/adult.html.
- Jones LA, Brierley DI. GLP-1 and the Neurobiology of Eating Control: Recent Advances. Endocrinology. 2025 Jan 6;166(2):bqae167. doi: 10.1210/endocr/bqae167.
- Jastreboff AM, Kotz CM, Kahan S, Heymsfield SB. Obesity as a disease: the obesity society 2018 position statement. Obesity (Silver Spring). 2019;27(1):7-9. doi:10.1002/oby.22378.
- American Diabetes Association Professional Practice Committee for Diabetes*. 8. Obesity and Weight Management for the Prevention and Treatment of Diabetes: Standards of Care in Diabetes-2026. Diabetes Care. 2026 Jan 1;49(Supplement_1):S166-S182. doi: 10.2337/dc26-S008.
- American College of Cardiology. Obesity and cardiovascular disease: updated clinical guidance. J Am Coll Cardiol. 2025 Aug 19;86(7):536-555. doi: 10.1016/j.jacc.2025.05.024.doi:10.1016/j.jacc.2025.05.024.
- Sievenpiper JL, Ard J, Blüher M, et al. Nutritional and lifestyle supportive care recommendations for management of obesity with GLP-1–based therapies: an expert consensus statement using a modified Delphi approach. Obes Pillars. 2025;17:100228. doi:10.1016/j.obpill.2025.100228.
- Bays HE, Toth P, Alkhouri N, et al. Weekly subcutaneous VK2735, a GIP/GLP-1 receptor dual agonist, for weight management: phase 2, randomized, 13-week VENTURE study. Obesity (Silver Spring). 2026. doi:10.1002/oby.70106. https://onlinelibrary.wiley.com/doi/10.1002/oby.70106.
- Viking Therapeutics, Inc. VK2735 for Weight Management Phase 3 (VANQUISH-1). ClinicalTrials.gov Identifier: NCT07104500. Updated November 24, 2025. Accessed February 26, 2026. https://clinicaltrials.gov/study/NCT07104500.
- Viking Therapeutics, Inc. VK2735 for Weight Management Type 2 Diabetes Phase 3 (VANQUISH-2). ClinicalTrials.gov Identifier: NCT07104383. Updated November 20, 2025. Accessed February 26, 2026. https://clinicaltrials.gov/study/NCT07104383.
